There is growing evidence that phytochelatins may be important for metal detoxifications not only in plants but also in many different animal species.
Background:How do animals respond to environmental pollution by potentially toxic elements (PTEs) such as heavy metals, what detoxification pathways do they use, how can the different sensitivity and vulnerability of different species be explained ?
These are key questions, not only to understand the fundamental biological responses of animals exposed to toxic metal concentrations in contaminated environments, but also to assess if these responses can have practical uses, for example in environmental biomonitoring.
Metallothioneins (MT) have been especially widely studied in this context.
Metallothioneins are small cysteine-rich proteins that strongly bind
soft metal ions such as cadmium. These proteins have been isolated and studied in a wide variety of organisms, including prokaryotes, plants, invertebrates and vertebrates. The metal binding domain of MT consists of 20 cysteine residues, whereby the sulfhydryl groups can bind 7 moles of divalent metal ions per mol of MT, while the molar ratio for monovalent metal ions (Cu and Ag) is twelve. Although the naturally occurring protein has Zn(II) in both binding sites, this ion may be displaced by other metal ions that have a higher affinity for thiolate such as Pb(II), Cu(I), Cd(II), Hg(II), Ag(I), Pt(II and IV), and/or Pd(II). MTs have repeatedly been shown to be strongly metal-inducible in many
different animal species, and there is ample evidence (again, in
multiple species) that knocking out MTs reduces tolerance
to heavy metals such as cadmium.
However, it is also clear that MTs are not the sole players in detoxification, and that MTs have many biological roles beyond detoxification including metal storage and transport and in metal ion homeostasis. As a result, the baseline variability of metallothioneins in the natural environment can be high, which can also complicate their use as biomarkers of pollution.
So the question is, what other biological systems are involved in responses to toxic elements ? A group of researchers from the UK now summarized the existing information on the role of phytochelatins and concluded that there is growing evidence that phytochelatins may be important not only in microorganisms and plants but also in many different animal species.
The role of Phytochelatins for detoxification of heavy metals: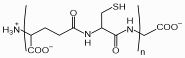
Chemical structure of phytochelatins
with n = 2-11.
|
Phytochelatins, like metallothioneins, are cysteine-rich peptides;
unlike metallothioneins, they are not genetically encoded, but are
nonribosomal peptides produced from glutathione by the enzyme
phytochelatin synthase (PCS). Originally thought to be found only in plants and yeast, PCS genes have since been found in species that span almost the whole animal tree of life (with some important exceptions).
The group of researchers argues that studying phytochelatin responses in animal species and their interactions with metallothioneins should be an important future goal. PCS genes have been found already in animal species from eight phyla, including species that are widely used in environmental toxicity testing and environmental monitoring, such as the oyster Crassostrea gigas, as well as nematodes and earthworms.
The group of researchers from the UK proposes four questions for future research:
- Which animal species with PCS genes make phytochelatins in response to toxic metal/metalloid ions ?
- What happens to metal ions once they have been bound by phytochelatins?
- Do phytochelatins interact with metallothioneins to help detoxify PTEs?
- And could phytochelatin levels potentially be used as biomarkers of environmental pollution?
The researchers expressed their opinion that answering these questions would be an important step forward in understanding how pollution by PTEs affects key invertebrate species in the environment.
Interesting work ahead for scientists working in speciation analysis !
 The cited viewpoint
The cited viewpoint
Jacob G. Bundy, Peter Kille, Manuel Liebeke, David J. Spurgeon,
Metallothioneins May Not Be EnoughThe Role of Phytochelatins in Invertebrate Metal Detoxification, Environ. Sci. Tehnol., 48/2 (2014) 885-886. DOI: 10.1021/es4054779
 Related studies
Related studies (newest first)

C. Rigouin, E. Nylin, A.A. Cogswell,
D. Schaumlöffel, D. Dobritzsch, D.L. Williams,
Towards an understanding of the function of the phytochelatin synthase of Schistosoma mansoni, PLoS Neglected Trop. Dis., 7 (2013) e2037.
DOI: 10.1371/journal.pntd.0002037
M. Liebeke, I. Garcia-Perez, C.J. Anderson, A.J. Lawlor, M.H. Bennett, C.A. Morris, P. Kille, C. Svendsen, D.J. Spurgeon, J.G. Bundy,
Earthworms produce phytochelatins in response to arsenic, PLoS One, 8 (2013) e81271.
DOI: 10.1371/journal.pone.0081271

B. Alan Wood,
Jörg Feldmann,
Quantification of phytochelatins and their metal(loid) complexes: critical assessment of current analytical methodology, Anal. Bioanal, Chem., 402 (2012) 3299-3309.
DOI: 10.1007/s00216-011-5649-0
S.L. Hughes, J.G. Bundy, E.J. Want, P. Kille, S.R. Sturzenbaum,
The metabolomic responses of Caenorhabditis elegans to cadmium are largely independent of metallothionein status, but dominated by changes in cystathionine and phytochelatins, J. Proteome Res., 8 (2009) 3512 3519.
DOI: 10.1021/pr9001806
J.C. Amiard, C. Amiard-Triquet, S. Barka, J. Pellerin, P.S. Rainbow,
Metallothioneins in aquatic invertebrates: Their role in metal detoxification and their use as biomarkers, Aquat. Toxicol., 76 (2006) 160202. DOI:

Masahiro Inouhe,
Toxic Metals in Plants: Phytochelatins, Braz. J. Plant Physiol., 17/1 (2005) 65-78.
DOI: 10.1590/S1677-04202005000100006 
C. Cobbett, P. Goldsbrough,
Phytochelatins and metallothioneins: roles in heavy metal detoxification and homeostasis, Annu. Rev. Plant Biol., 53 (2002) 159 182.
DOI: 10.1146/annurev.arplant.53.100301.135154 Related EVISA Resources
Related EVISA Resources Brief summary: ICP-MS - A versatile detection system for speciation analysis
Brief summary: ICP-MS - A versatile detection system for speciation analysis Brief summary: LC-ICP-MS - The most often used hyphenated system for speciation analysis
Brief summary: LC-ICP-MS - The most often used hyphenated system for speciation analysis  Link page: Mass spectrometry resources
Link page: Mass spectrometry resources Glossary: Phytochelatins
Glossary: Phytochelatins
last time modified: January 31, 2014