Organoarsenic compounds were found in some low rank coals from Slovenia and the Czech Republic. Most likely biogeochemical degradation of organic material is responsible for these unexpected organoarsenic compounds.
The origin of coal
Coal is a result of the accumulation and slow decay of plant remains in sedimentary strata. It undergoes in situ compaction under water with time, accompanied by biochemical processes such as decomposition due to bacterial action, dehydration, loss of volatile compounds (e.g. methane, higher hydrocarbons, carbon dioxide and nitrogen) and densification to form various ranks of coal depending on environmental conditions. In absence of atmospheric oxygen, the plant matter is further degraded by the action of anaerobic bacteria, which extract and utilize oxygen from organic molecules containing oxygen like lignin.
Where does the arsenic come from ?
Arsenic, one of the potentially hazardous trace elements, is usually concentrated in the sulphidic minerals of coal. Many authors interpret the arsenic accumulation in terms of arsenic concentration during decay of plant matter in the humic layers. The arsenic content of coal samples worldwide is highly variable, with an average value around 5 mg kg-1 and extreme high values of up to 35,000 mg kg-1 in coals from endemic arsenosis areas in China.
In which form is arsenic present in fossil fuels ?
Data on the form of arsenic in coal are scarce. The group of Finkelman [1] examined polished blocks of coal using a scanning electron microscope with an energy-dispersive X-ray analyser (SEM-EDS) and an electron microprobe (EMPA). They found a wide variety of arsenic bearing minerals with pyrite being the most common one. Pyrite was the main arsenic form in four US bituminous coals studied by X-ray absorption fine structure (XAFS) for arsenic speciation nalysis, although arsenate and arsenic associated with organic matter were found as well [2].
On the other hand, there are some data on organoarsenic compounds in other fossil fuels. Phenylarsonic acid, monomethyl arsonic acid (MMAA) and dimethylarsinic acid (DMAA) were identified in oil shales and process waters [3] and the author explained the origin of these compounds by either biological synthesis in the original organisms which formed oil (bioaccumulation), or by purely abiotic synthesis in the hot retort during the processing of oil. Also volatile organoarsenic species such as trimethylarsine, dimethylarsine, methyldiethylarsine and triethylarsine were found in natural gas samples [4].
Low rank coals from Slovenia and Czech Republic
In our study low rank coal samples from three different coal basins (Velenje and Trbovlje basins, Slovenia and Sokolov basin, Czech Republic) were analysed for the presence of arsenic compounds [5]. Total arsenic concentrations in the samples were 1.6-5.8 µg g-1 with one exception, 142 µg g-1 for a sample from the Sokolov basin. Extracts from the Velenje basin samples contained mainly the tetramethylarsonium ion (TETRA) with considerable amounts of arsenate and monomethyl arsonic acid (MMAA) while in the samples from the Sokolov and Trbovlje basins, inorganic arsenic prevailed over organic arsenic compounds see figure below).
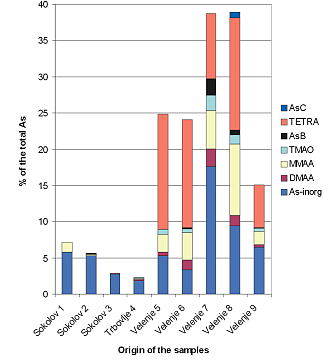
Although the presence of organically bound arsenic was suggested earlier for some Australian coals [6], this is the first report on the presence of the organoarsenic compounds MMAA, DMAA, TETRA, TMAO and AsB in coal samples [5]. The non-extractable fraction of arsenic, probably mineral phases such as arsenopyrite etc., was not further investigated.
Figure 1 Arsenic speciation in extracts of coal samples as determined by HPLC-UV-HGAFS
Where are the organoarsenic compounds coming from ?
Since organic arsenic compounds are characteristic of living organisms, the question arises how they emerged in coal samples. Organic matter in coal originates from plants, which in general contain very little arsenic. Although arsenic compounds are rather stable under laboratory conditions, the stability of organic arsenic compounds under the harsh conditions during coal genesis is rather unlikely - even minor decomposition would result in total degradation of organoarsenic compounds over a period of millions of years. Purely abiogenic synthesis of organoarsenic compounds in an environment very rich in carbon in combination with long time periods and elevated temperatures can also not be excluded as suggested by Fish [3] for an oil shale environment.
The most probable explanation seems to us the microbial synthesis of organoarsenic compounds. In all three basins investigated the presence of microorganisms, especially methanogenic ones, is documented [7]. Petrographic and geochemical data of gelified and ungelified fossil wood provide evidence that gelification may be governed by microorganisms like anaerobic bacteria. Therefore it is possible that the presence of organic arsenic compounds is related to the bacterial activity that took place (and is still occurring) in the formation of lignite, especially in Velenje, but also in the other two coal seams studied. Bacteria have been found in some coal samples and in sludge, in which they were able to produce volatile metal(loid) derivatives as documented by Michalke et al [8] for some methanogenic and other bacteria, which were able to produce volatile methylated arsenic, antimony, bismuth, selenium or tellurium compounds.
Zdenka lejkovec and Tjaa Kanduč
Joef Stefan Institute, Jamova 39, 1000 Ljubljana, Slovenia
 The original study:
The original study:

Zdenka lejkovec and Tjaa Kanduč,
Unexpected Arsenic Compounds in Low-Rank Coals, Environ. Sci. Technol., 39/10 (2005) 34503454.
DOI: 10.1021/es0400990
 Cited studies
Cited studies

[1] R.B. Finkelman, W. Orem, V. Castranova, C.A. Tatu, H.E. Belkin, B. Zheng, H.E. Lerch, S.V. Maharaj and A.L. Bates,
Health impacts of coal and coal use: possible solutions, Int. J. Coal Geol., 50 (2002) 425-443.
DOI: 10.1016/S0166-5162(02)00125-8

[2] R.H. Fish,
Fingerprinting inorganic arsenic and organoarsenic compounds in In-Situ oil shale retort and process waters using a liquid chromatograph coupled with an atomic absorption spectrometer as detector, Environ. Sci. Technol., 16 (1982) 174.
DOI: 10.1021/es00097a010

[3] A. Kolker, F.E. Huggins, C.A. Palmer, N. Shah, S.S. Crowley, G.P. Huffman and R.B. Finkelman,
Mode of occurrence of arsenic in four US coals, Fuel Process. Technol., 63 (2000) 167-178.
DOI: 10.1016/S0378-3820(99)00095-8

[4] K.J. Irgolic, D. Spall, B.K. Puri, D. Ilger and R.A. Zingaro,
Determination of arsenic and arsenic compounds in natural gas samples, Appl. Organomet. Chem., 5 (1991) 117-124.
DOI: 10.1002/aoc.590050209

[5] Z. lejkovec and T. Kanduc,
Unexpected Arsenic Compounds in Low-Rank Coals, Environ. Sci. Technol., 39 (2005) 3450-3454.
DOI: 10.1021/es0400990

[6] V. Valkovic, Trace elements in coal, CRC Press: Boca Raton, 1983.

[7] T. Kanduc and J. Pezdic, Geochemical Journal, in press.

[8] K. Michalke, E.B. Wickenheiser, M. Mehring, A.V. Hirner and R. Hensel,
Production of volatile derivatives of metal(loid)s by microflora involved in anaerobic digestion of sewage sludge, Appl. Environ. Microbiol., 66 (2000) 2791.
DOI: 10.1128/AEM.66.7.2791-2796.2000
 Related information about coal
Related information about coal
 The Society of Organic Petrology
The Society of Organic Petrology
 Related information about arsenic in coal
Related information about arsenic in coal

J. Liu, B. Zheng, H.V. Aposhian, Y. Zhou, M.L. Chen, A. Zhang, M.P. Waalkes,
Chronic arsenic poisoning from burning high-arsenic-containing coal in Guizhou, China, Environ Health Perspect., 110/2 (2002) 119-22.
DOI:
10.1289/ehp.02110119

F.H. Zhao, D.Y. Ren, D.W. Xu, et al.,
Research on the phase of arsenic in coal-burning residue, Journal of China University of Mining & Technology, 28/4 (1999) 365367.

J.P. Zhang, Y.Q. Wang, R.G. Zhang, et al.,
Distribution of arsenic in coal and its residues, Research of Environmental Sciences, 12/1 (1999) 2729,34.

F.H. Zhao, D.Y. Ren, S.P. Peng, et al.,
The modes of occurrence of arsenic in coal, Advance in Earth Science, 18/2 (2003) 214220.
DOI: 10.1007/BF02883414

Y.P. Zhou,
Distribution type and occurrence form of arsenic in anthracite of Laochang mining area, Coal Geology & Exploration, 26/4 1998) 813.
 Related (EVISA) News
Related (EVISA) News November 14, 2013: Arsenic speciation in rice cereals for infants
November 14, 2013: Arsenic speciation in rice cereals for infants May 15, 2013: Arsenic species in rice: Origin, uptake and geographical variation
May 15, 2013: Arsenic species in rice: Origin, uptake and geographical variation September 21, 2012: Arsenic in Rice : First results from the U.S. Food and Drug Administration
September 21, 2012: Arsenic in Rice : First results from the U.S. Food and Drug Administration  January 4, 2011: Arsenic species in rice: Call for analytical laboratories
January 4, 2011: Arsenic species in rice: Call for analytical laboratories  August 2, 2010: Gut bacteria transform inorganic arsenate leading to more toxic arsenic species
August 2, 2010: Gut bacteria transform inorganic arsenate leading to more toxic arsenic species  May 19, 2010: China: Inorganic Arsenic in Rice - An Underestimated Health Threat ?
May 19, 2010: China: Inorganic Arsenic in Rice - An Underestimated Health Threat ?  February 23, 2010: US EPA opens inorganic arsenic cancer assessment for public review
February 23, 2010: US EPA opens inorganic arsenic cancer assessment for public review December 4, 2009: EFSA: Scientific Opinion on Arsenic in Food
December 4, 2009: EFSA: Scientific Opinion on Arsenic in Food May 26, 2009: UK Food Standards Agency releases research on arsenic in rice milk
May 26, 2009: UK Food Standards Agency releases research on arsenic in rice milk  January 31, 2009: Using the right recipe for cooking rice reduces toxic inorganic arsenic content
January 31, 2009: Using the right recipe for cooking rice reduces toxic inorganic arsenic content November 11, 2008: EFSA calls for data on arsenic levels in food and water
November 11, 2008: EFSA calls for data on arsenic levels in food and water September 5, 2008: Exposure to inorganic arsenic may increase diabetes risk
September 5, 2008: Exposure to inorganic arsenic may increase diabetes risk March 15, 2008: Arsenic in rice milk exceeds EU and US drinking water standards
March 15, 2008: Arsenic in rice milk exceeds EU and US drinking water standards February 15, 2008: Arsenic speciation in rice: a question of the rice plant species
February 15, 2008: Arsenic speciation in rice: a question of the rice plant species December 26, 2007: The effect of thermal treatment on the arsenic speciation in food
December 26, 2007: The effect of thermal treatment on the arsenic speciation in food  March 7, 2007: Elevated Arsenic Levels Found In Rice Grown In South Central States of the USA
March 7, 2007: Elevated Arsenic Levels Found In Rice Grown In South Central States of the USA
 January 18, 2006: Hungarians exposed to high arsenic levels in drinking wate
January 18, 2006: Hungarians exposed to high arsenic levels in drinking water
 August 3, 2005: Surprisingly high concentrations of toxic arsenic species found in U.S. rice
August 3, 2005: Surprisingly high concentrations of toxic arsenic species found in U.S. rice  May
15, 2005: Use of organoarsenicals as pesticides may lead to
contamination of soils and groundwater with toxic arsenic species
May
15, 2005: Use of organoarsenicals as pesticides may lead to
contamination of soils and groundwater with toxic arsenic species
last time modified: March 6, 2024