A group of French researchers developed a method for enhancing the poor extraction of retained gadolinium from brain tissue. Subsequent analysis using size exclusion chromatographyinductively coupled plasmamass spectrometry (SEC-ICP-MS) was intended to shed the light on the speciation of the additionally recovered Gd.
Background:Gadolinium-based contrast agents (GBCAs) are used since their introduction in the 1980s for improving the contrast in magnetic resonance imaging for medical diagnostics. In order to reduce the toxicity of the paramagnetic gadolinium (Gd), the metal is complexed with polyaminocarboxylic acid. These chelates are selected in order to create complexes of relatively high thermodynamic and kinetic stability and high water solubility. As a result, the GBCAs are in general excreted after administration unmetabolized to great extent in relatively short times. Related to the different chelates used, the GBCAs can be divided into linear and macrocyclic ones, either class of which can be further subdivided into ionic and non-ionic compounds. Under physiological conditions, the macrocyclic GBCAs are more stable (especially kinetic wise) than the linear type and the ionic linear GBCAs are more stable than the non-ionic linear GBCAs.
While considered to be safe for nearly 25 years, in the last decade it has become well established that a trace amount of Gd does accumulate for a prolonged time in multiple organs, including the brain, after a single and even more so after repeated GBCAs administration. Since the degree of retention observed was found to be more pronounced for linear GBCAs, the European Medicines Agency (EMA) has retracted the approval of linear GBCAs for general application in Europe.
While the retention of Gd in different tissues could not be related to clinical effects, a complete understanding of the retention mechanism and the characterization of the Gd species involved, is essential for the long-term GBCA safety. Numerous studies have tried to shed light on this issue, with many questions left open. Several studies have shown that Gd retention is more pronounced with linear GBCAs than with macrocyclic ones. The distribution of Gd in different tissues is not uniform, but shows quite high heterogeneity, with hot spots in areas rich in endogenous metals such as iron. Such findings have fueled the hypothesis, that transmetallation may withdraw the Gd from the chelate, making the Gd available for binding with biomolecules. A multimodal imaging study using transmission electron microscopy (TEM) associated with nanoSIMS (nanoscale secondary ion mass spectrometry) demonstrated the presence of Gd deposits containing phosphorus in the deep cerebellar nuclei (DCN), strongly suggesting formation of GdPO4. While such hypothesis is only based on co-localization, more direct identification of the Gd-species directly in the tissue by using synchrotron-based techniques such as EXAFS and XANES were not successful up to now.
Alternative techniques based on the use of chromatographic species separation and identification by mass spectrometry require having the species extracted into solution. Here the problem is to obtain high extraction efficiency without harsh conditions that would destroy the original species. Most of the extraction procedures reported could only extract a minor part of the retained Gd, leaving the question of the retained species open.
The new studyA group of French researchers aimed at the development of a method able to solubilize the brain tissue completely and to obtain an insight into the speciation of the additionally recovered Gd in relation to the GBCA administered.
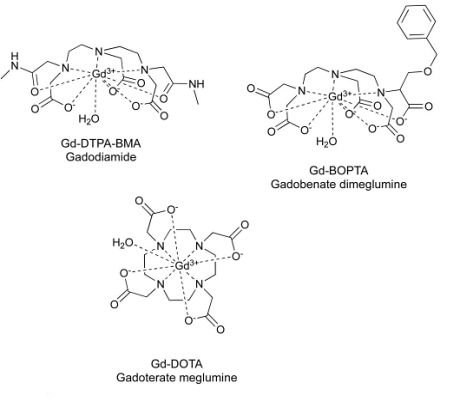
To cover the different GBCA classes available on the market, three GBCAs were selected for the study (1 macrocyclic and ionic GBCA gadoterate meglumine and 2 linear [1 ionic gadobenate dimeglumine and 1 nonionic gadodiamide]). The GBCAs were administered to healthy female rats once per week during 5 weeks at a dose of 2.4 mmol/kg (corresponding to 0.4 mmol/kg in humans). The animals were sacrificed 1 week after the last injection. The brains were removed and dissected to sample a half cerebellum, half cortex, and half subcortical brain. Tissue samples were stored at -80°V before further sample preparation.
For extraction, the tissue samples were homogenized in 100 mM ammonium acetate by using an Ultra-Turrax disperser. A sequential extraction procedure using water in the first step, urea in the second step was applied. The final urea-insoluble residue was washed three times with water. For total Gd determination, samples of tissue homogenate were digested with HNO3/H2O2 at 80°C for 8 hours. Total Gd concentrations were measured by ICP-MS with external calibration in the range of 0.010 to 1.0 ng/mL. The concentration in the cerebellum obtained after injections of rats with gadoterate (0.63 ± 0.12 nmol/g) was lower than that obtained after injections with either linear contrast agents: gadobenate (1.52 ± 0.16 nmol/g) and gadodiamide (2.74 ± 0.10 nmol/g). The same trend was observed for other tissues.
The water extraction allowed the recovery of 60-72% of Gd initially present in the tissue of animals treated with the macrocyclic gadoterate. For linear GBCAs, the extracted amounts reached only 13-16% for gadobenate and 12-13% for gadodiamide. Extraction efficiency was significantly improved after solubilization with urea, reaching 88+-1 3% for gadoterate, 47 ± 2% for gadobenate and 44 ± 27% for gadodiamide. Only a very small amount of Gd (1-2%) remained in the final insoluble residue.
For speciation analysis, size-exclusion chromatography was coupled with ICP-MS. Stability test of the three GBCAs with control samples indicated that no degradation was occurring during analysis. Also, column recovery was quantitative for all three GBCAs.
In the water extract of samples originating from animals treated with the macrocyclic gadoterate, only the intact GBCA was found. In the samples from animals treated with linear GBCAs 3 distinct peaks were observed. While the last one was belonging to the original GBCAs, the two earlier peaks belong to compounds greater than 81 kDa and greater than 66 kDa. Also the column recovery for samples of linear GBCAs was only 82 ± 2%, suggesting that a no negliable amount of Gd present in the extract was lost
Also, the urea extraction procedure was tested for degradation problems. There was no indication for degradation for the three GBCAs and also column recovery was quantitative from spiked control samples. Again, as for water extracts, the macrocyclic gadoterate was extracted as the only species present. For the linear GBCAs, the chromatograms showed at least three peaks, with two of them belonging to compounds larger than 80 kDA.
Using another SEC column with higher separation range, the two peaks were classified as belonging to compounds larger than 660 kDa and one of approximately 440 kDa. Ferritin was identified as a potential endogenous macromolecule interacting with the dissociated Gd3+. Also, the poor column recovery indicated that about 38% of extracted Gd was retained on the column. Moreover, while urea extraction improved the extraction efficiency significantly, approximately half of the Gd remained in the insoluble residue for linear GBCAs.
The authors concluded that further investigations would be of interest in order to learn more about those unidentified Gd species retained in the extraction residue.
 The cited study:
The cited study:

Izabela Strzeminska, Cécile Factor, Philippe Robert,
Joanna Szpunar, Claire Corot,
Ryszard Lobinski, Speciation Analysis of Gadolinium in the Water-Insoluble Rat Brain Fraction After Administration of Gadolinium-Based Contrast Agents, Invest. Radiol., 56/9 (2021) 535-544.
DOI: 10.1097/RLI.0000000000000774 Used Instrumentation:
Used Instrumentation:
 Agilent Technologies - 7700x ICP-MS
Agilent Technologies - 7700x ICP-MS Agilent Technologies - 1260 HPLC system
Agilent Technologies - 1260 HPLC system
 Related studies (newest first):
Related studies (newest first):
I. Strzeminska, C. Factor, P. Robert, A.-L. Grindel, P.-O. Comby,
J. Supunar, C. Corot,
R. Lobinski, Long-term evaluation of gadolinium retention in rat brain after single injection of a clinically relevant dose of gadolinium-based contrast agents. Invest. Radiol. 55 (2020) 138143.
DOI: 10.1097/RLI.0000000000000623
A.A.P. Kartamihardja, H. Hanaoka, P. Andriana, S. Kameo, A. Takahashi, H. Koyama H, Y. Tsushima,
Quantitative analysis of Gd in the protein content of the brain following single injection of gadolinium-based contrast agents (GBCAs) by size exclusion chromatography. Br. J. Radiol., 92 (2019) 20190062.
DOI: 10.1259/bjr.20190062
P. Robert, S. Fingerhut, C. Factor, V. Vives, J. Letien,
M. Sperling, M. Rasschaerts, R. Santus, S. Ballet, Jean-Marc Idée, C. Corot,
U. Karst,
One-year retention of gadolinium in the brain: comparison of gadodiamide and gadoterate meglumine in a rodent model. Radiology., 288 (2018) 424433.
DOI: 10.1148/radiol.2018172746
T. Frenzel, C. Apte, G. Jost, L. Schöckel, J. Lohrke, H. Pietsch,
Quantification and assessment of the chemical form of residual gadolinium in the brain after repeated administration of gadolinium-based contrast agents: comparative study in rats. Invest Radiol., 52 (2017) 396404.
DOI: 10.1097/RLI.0000000000000352
M. Birka, K.S. Wentker, E. Lusmöller, B. Arheilger, C.A. Wehe,
M. Sperling, R. Stadler,
U. Karst,
Diagnosis of Nephrogenic Systemic Fibrosis by means of Elemental Bioimaging and Speciation Analysis, Anal. Chem., 87 (2015) 3321−3328.
DOI: 10.1021/ac504488k
L. Telgmann,
M. Sperling,
U. Karst,
Determination of gadolinium-based MRI contrast agents in biological and environmental samples: A review, Anal. Chim. Acta, 764 (2013) 116.
DOI: 10.1016/j.aca.2012.12.007
 Related EVISA News (newest first)
Related EVISA News (newest first)
last time modified: January 12, 2025