Using a commercial platform automation system, scientists from Elemental Scientific have developed methods for the total arsenic determination and its speciation in urine samples. The obtained results with these methods demonstrate high sensitivity, high sample throughput and excellent accuracy.
Background:It is common understanding that arsenic is an important health hazard for humans, with a toxicity that heavily depends on the arsenic species being present. Arsenic can be found in the biosphere in many different forms; the number of identified species has surpassed one hundred. The inorganic forms, arsenite (As(III)) or arsenate (As(V)), are more toxic due to higher bioavailability. Some organic forms (dimethylarsinic acid (DMA) and monomethylarsonic acid (MMA)) are less toxic due to a lower bioavailability, while other organic forms of arsenic such as arsenobetaine (AsB), arsenocholine (AsC), and trimethylarsine oxide (TMAO) are considered non-toxic. However, unfortunately also some organic arsenic species have been identified, that show an important toxicity comparable to that of inorganic species. Human exposure routes are mainly from drinking water or food consumption. Arsenic speciation analysis is therefore a prerequisite for meaningful risk assessment, as well as for the study of metabolic processes or its biogeochemical cycling in the environment. The most common methodology for arsenic speciation analysis is based on the separation of species by liquid chromatography (LC) followed by detection by inductively coupled plasma mass spectrometry (ICP-MS). Since the additional separation of species is reducing the sample throughput of the hyphenated technique LC-ICP-MS in comparison to the total element concentration measurement by ICP-MS, the LC-ICP-MS approach is more complex and costlier. The development of methods for such analysis is therefore aiming at efficient separation of the target species in a minimum of time.
The new methods:Scientists from the company Elemental Scientific have developed methods for total arsenic determination and arsenic speciation analysis based on the use of a single platform automation system (prepFast IC) connected to a single ICP-MS. The hyphenated system provides automation in the sample preparation and delivery, but also reduces the potential bias of having two completely different setups for these measurements. Two different arsenic speciation methods were developed and evaluated for AsB, AsC, DMA, MMA, As(III), As(V), and TMAO. The methods were evaluated for column recovery, accuracy, precision, and limits of detection. Accuracy of the methods were evaluated by analysing proficiency testing samples from the Centre de Toxicologie du Québec (CTQ) and New York Department of Health (NYDOH). Synthetic clinical matrix (CLIN-0500, Elemental Scientific) was used to matrix-match the calibration curves for direct mode analysis of urine samples; no matrix-matching was used for the urine As speciation analysis.
Calibration curves for total arsenic were prepared by using stock solutions containing 100 µg/L As. Calibration curves for the arsenic speciation were prepared using a stock standard of 100 µg/L of each of the arsenic species. Samples to be analysed were proficiency testing (PT) samples obtained from the Centre de Toxicologie du Québec (CTQ) and New York Department of Health (NYDOH)as well as human urine samples spiked for individual As species.
Standards and samples were introduced to the ICP-MS using a prepFast IC Clinical system. The prepFast IC is an automated total metals and chromatography system, which includes an autosampler and the ability to dilute standards and samples inline to the ICP-MS. The prepFAST IC system can operate in total metals or elemental speciation modes by either passing the sample through a separation column or bypassing the column. The system was configured with an anion exchange column (column valve A) and a C18 column (column valve B).
The ICP-MS was operated in KED mode with He in the octopole collision cell. Ammonium carbonate was selected as the eluent for the separation of AsB, As(III), DMA, AsC, MMA, and As(V). This separation was optimized to ~2 minutes using a gradient step of 0.5 mM ammonium carbonate followed by 80 mM ammonium carbonate with an anion exchange column. In this separation, AsB and TMAO both eluted in the void volume (Fig. 1a). Thus, if distinguishing between AsB and TMAO is critical a second method is required. Fig. 1b displays the separation of AsB, TMAO, As(III), DMA, AsC, MMA, and As(V) performed using the combination of an anion exchange column and a C18 column.
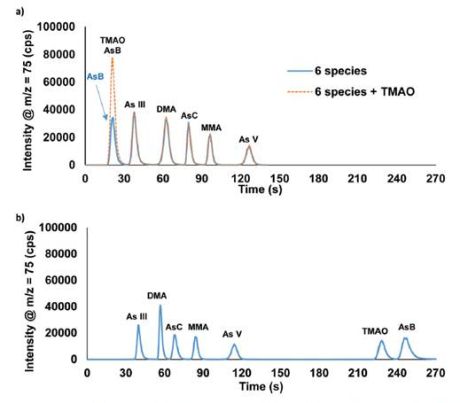
Figure 1: Chromatographic separation of AsB, TMAO, As(III), DMA, AsC, MMA, and As(V) using the (a) one-column and (b) two-column methods. Each species was spiked into the urine sample at 50 μg L−1 As.
The separation is done by sending the sample through the anion exchange
column first; the AsB/TMAO peak elutes off of the column and passes into
the C18 column. Both columns are on switching valves, which allows for
the C18 column to be then switched offline at this point in the method,
trapping the AsB and TMAO species. The As(III), DMA, AsC, MMA, and As(V)
are eluted off of the anion exchange column, followed by a switch back
to eluent 1 and the C18 column brought back online. Eluent 1 is then
used to elute TMAO and AsB off of the C18 column. The total separation
time for the two-column method was optimized to ~4.5 minutes.
Both methods were evaluated for recovery of 10 µg/l arsenic spiked as individual species to human urine. Both methods had very good recovery that ranged between 94 and 105%. The detection limits were evaluated from blank readings and ranged between 3 and 6 ng/L for the one-column method and 4 and 9 ng/L for the two-column method. The methods were also validated by analysing proficiency test samples from CTQ and NYDOH. There was excellent correlation between results and target values, showing up in a linear regression slope of 0.9938.
 The original study
The original study
 C. Derrick Quarles, Jr
C. Derrick Quarles, Jr, Patrick Sullivan, Nick Bohlim and Nathan Saetveit,
Rapid automated total arsenic and arsenic speciation by inductively coupled plasma mass spectrometry, J. Anal. At. Spectrom., 37/6 (2022) 1249-1246.
DOI: 10.1039/d2ja00055e
 Instrumentation used:
Instrumentation used:
 Agilent Technologies - 7900 ICP-MS
Agilent Technologies - 7900 ICP-MS Elemental Scientific - prepFAST IC
Elemental Scientific - prepFAST IC
 Related studies (newest first)
Related studies (newest first)
P.F. Rodriguez, R.M. Martin-Aranda, J.L. Lopez Colon, J.H. de Mendoza,
Ammonium acetate as a novel buffer for highly selective robust urinary HPLC-ICP-MS arsenic speciation methodology, Talanta, 2021, 221 , 121494.
DOI: 10.1016/j.talanta.2020.121494
X. Song, L. Huiling, C. Ma, W. Duan, Y. Sun, Y. Li, T. Huang, B. Zhou,
Clinimetallomics: Arsenic Speciation in Urine from Patients with Arsenism by HPLC-ICP-MS. At. Spectrosc., 2021, 42 , 278 281.
DOI: 10.46770/AS.2021.103

P. Montoro-Leal, J.C. García-Mesa, I. Morales-Benitez, A. Garcia de Torres, E. Vereda Alonso,
Semiautomatic method for the ultra-trace arsenic speciation in environmental and biological samples via magnetic solid phase extraction prior to HPLC-ICP-MS determination, Talanta, 2021, 235 , 122769. DOI: 10.1016/j.talanta.2021.122769

P.J. Wegwerth, S.A. Erdahl, M.L. Wermers, M.M. Hanley, S.J. Eckdahl, P.J. Jannetto,
Reliable, Rapid, and Robust Speciation of Arsenic in Urine by IC-ICP-MS , J. Appl. Lab. Med., 2021, 846 857.
DOI: 10.1093/jalm/jfaa226 C.D. Quarles
C.D. Quarles, P. Sullivan, M.P. Field, S. Smith, D.R. Wiederin,
Use of an inline dilution method to eliminate species interconversion for LC-ICP-MS based applications: focus on arsenic in urine, J. Anal. At. Spectrom., 2018, 33 , 745 751.
DOI: 10.1039/C8JA00038G
 Related EVISA News (Newest first)
Related EVISA News (Newest first)
last time modified: January 11, 2025